Skin-deep smell receptors speed up healing process
Skin-deep smell receptors speed up healing process
November 3, 2014
Olfactory receptors—the type of receptors in the human body responsible for the sense of smell—were first detected outside the nose in 2003. They were found in sperm cells, to be exact. Since then, these receptors which, were once thought to only play a role in the sense of smell, have been identified in a variety of human tissues, including the intestines, kidneys and prostate.
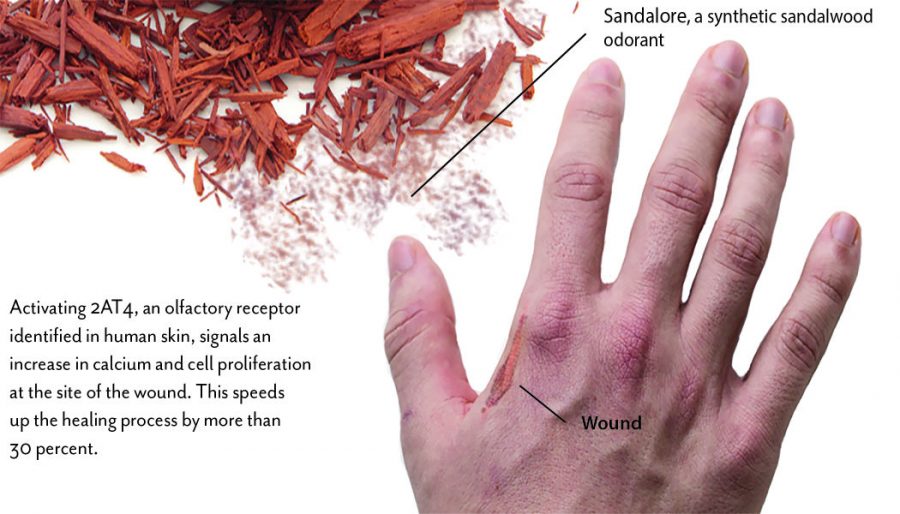
A study published Aug. 7 in the Journal of Investigative Dermatology found a new olfactory receptor in skin cells that speeds up the healing process by more than 30 percent in the presence of a synthetic sandalwood odorant called Sandalore, which the cells were exposed to in a test tube for five days.
All cells contain genes that are either activated or lie dormant, determining the function of that cell. Scientists have developed methods for identifying the expression of these olfactory receptors in cells where they would not expect to find them, but identifying their functions is a more difficult task, according to Dr. Hanns Hatt, co-author of the study and a professor in the Department of Cell Physiology at Ruhr-Universität Bochum in Germany.
“We know exactly which receptors exist in which tissues, but the problem is that even though we can tell that a particular olfactory receptor may be highly expressed in lung tissue, we don’t know the odorant that can activate it,” Hatt said. “Without that odorant, we can’t study [its] function.”
The interaction between olfactory receptors and odor molecules works like a system of locks and keys, Hatt said. There are locks, or receptors, that can be opened with different keys, or odorants. Some locks only open with a certain key and some keys will open many locks, he added. A broadly tuned receptor can be activated by many odorants, but so far, in the case of the novel olfactory receptor protein, only a few chemical odorants have been found to activate it, making it narrowly tuned. Researchers also face the problem of possibly labeling a receptor narrowly tuned when it could be that other odorants that would activate it are not yet at their disposal.
Stuart Firestein, a professor in the Department of Biological Sciences at Columbia University, said olfactory receptors belong to a much larger family of receptors found all over the body and are capable of recognizing small molecules and reacting to them.
“They recognize small molecules when they’re inside our bodies or our brain in particular—things like neurotransmitters or hormones or other chemicals of some importance,” Firestein said. When they’re in the olfactory system, they recognize chemicals out in the world, out in the environment.”
Although Sandalore is a common chemical, often used to make synthetic sandalwood, researchers did not have a good understanding of which receptor would serve as its lock, Hatt said.
“We identified this receptor in the [skin cells] and found that Sandalore could stimulate it, resulting in a calcium increase,” Hatt said. “This calcium influx stimulates a biochemical cascade that leads to an increase in the proliferation of skin cells. The cells [multiply] faster and migrate to the wound.”
The research team used a technology called next-generation sequencing—which allowed them to profile which genes are expressed inside of a cell—to identify the novel smell receptor responsible for the accelerated healing.
Matthew Schipma, technical director of the Next Generation Sequencing Core facility at Northwestern University, said as genes within a cell are switched on or off, they manifest as transcripts, or messenger RNA. The mRNA is then isolated from the cell and turned into the type of DNA that is ultimately sequenced. Sequencing instruments generate hundreds of millions of DNA reads per cell.
“DNA reads are then mapped back to the genome,” Schipma said. “Gene expression is basically determined by how many reads align to a particular gene. This procedure gives scientists a way to estimate how active a gene is.”
Hatt said every cell in the body has the same number of about 20,000–25,000 genes, of which only about half are usually activated. Researchers were able to measure which of the approximately 350 olfactory receptor genes were most and least activated based on how many copies of that particular gene were found in a certain tissue.
“We know that in cancer cells, some of these olfactory receptor [proteins] are extremely highly expressed,” Hatt said. “There’s a receptor that exists in the prostate for the smell of viola that we can try to use as a diagnostic marker for cancerous cells. It also does the opposite of the [skin cell] receptor—this viola receptor stops the proliferation of cancer cells—so we know the olfactory receptor can have varying functions in different cell types.”